4.3: Mineral Identification
- Page ID
- 5363
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)How do you identify a mineral?
You're in a competition getting ready to go onstage. You discover that you forgot your shirt! You call your mom and ask her (nicely) to bring it to you right away. But first you need to let her know which shirt it is. What would you tell her? You might describe the color, the way the fabric feels, and the length of the sleeves. These are all physical properties of your shirt. If you do a good job describing the shirt your mom will bring the right one. Minerals also have physical properties that are used to identify them.
How Are Minerals Identified?
Imagine you were given a mineral sample similar to the one pictured below (Figure below). You can use a mineral’s properties to identify what type it is. How would you try to identify your mineral? You can observe some properties by looking at the mineral. For example, you can see that its color is green. The mineral has a needle-like, fibrous structure. But you can't see all mineral properties. You need to do simple tests to determine some properties. One common one is how hard the mineral is. The mineral’s physical properties are determined by its chemical composition and crystal structure.
Green actinolite.
Color, Streak, and Luster
Diamonds have many valuable properties. Diamonds are translucent and extremely hard. The most valuable diamonds are large, well-shaped and sparkly. Some are not as pretty and are used for industrial purposes. Turquoise is another mineral that is used in jewelry because of its striking greenish-blue color. Many minerals have interesting appearances. Specific terms are used to describe the appearance of minerals.
Color
Color is probably the easiest property to observe. Unfortunately, you can rarely identify a mineral only by its color. Sometimes, different minerals are the same color. For example, you might find a mineral that is a golden color and think it is gold. But it might actually be pyrite, or “fool's gold,” which is made of iron and sulfide. It contains no gold atoms.
A certain mineral may form in different colors. For example, pictured below are four samples of quartz (Figure below), including one that is colorless and one that is purple. The purple color comes from a tiny amount of iron. Iron is not normally found in quartz. When it is, it is a chemical impurity. Many minerals are colored by chemical impurities. Other factors can also affect a mineral’s color. Weathering changes the surface of a mineral. Because color alone is unreliable, geologists rarely identify a mineral just on its color. To identify most minerals, they use several properties.
Quartz comes in many different colors: (A) transparent quartz, (B) blue agate, (C) rose quartz, and (D) purple amethyst.
Streak
Streak is the color of the powder of a mineral. To do a streak test, you scrape the mineral across an unglazed porcelain plate. The plate is harder than many minerals, causing the minerals to leave a streak of powder on the plate. The color of the streak often differs from the color of the larger mineral sample (Figure below). Yellow-gold pyrite has a blackish streak. This blackish streak tells you that the mineral is not gold, because gold has a gold-colored streak.
Rub a mineral across an unglazed porcelain plate to see its streak. Hematite has a reddish-brown streak.
Streak is more reliable than color to identify minerals. Different minerals that are the same color may have a different color streak. For example, samples of hematite and galena are both dark gray. They can be told apart because hematite has a red streak and galena has a gray streak.
Luster
Luster describes the way light reflects off the surface of the mineral. You might describe diamonds as sparkly or pyrite as shiny. But mineralogists have special terms to describe luster. They first divide minerals into metallic and non-metallic luster. Minerals that are opaque and shiny, like pyrite, are said to have a “metallic” luster. Minerals with a “non-metallic” luster do not look like metals. There are many types of non-metallic luster. Six are described below (Table below).
| Non-Metallic Luster | Appearance |
|---|---|
| Adamantine | Sparkly |
| Earthy | Dull, clay-like |
| Pearly | Pearl-like |
| Resinous | Like resins, such as tree sap |
| Silky | Soft-looking with long fibers |
| Vitreous | Glassy |
Can you match the minerals (Figure below) with the correct luster, listed above (Table above), without looking at the caption?
(A) Diamonds have an adamantine luster. These minerals are transparent and highly reflective. (B) Kaolinite is a clay with a dull or earthy luster. (C) Opal’s luster is greasy. (D) Chalcopyrite, like its cousin pyrite, has metallic luster. (E) Stilbite (orange) has a resinous luster. (F) The white ulexite has silky luster. (G) Sphalerite has a submetallic luster. (H) Chert is a mineral with a waxy luster.
Density
You are going to visit a friend. You fill one backpack with books so you can study later. You stuff your pillow into another backpack that is the same size. Which backpack will be easier to carry? Even though the backpacks are the same size, the bag that contains your books is going to be much heavier. It has a greater density than the backpack with your pillow.
Density describes how much matter is in a certain amount of space. Substances that have more matter packed into a given space have higher densities. The water in a drinking glass has the same density as the water in a bathtub or swimming pool. All substances have characteristic densities, which does not depend on how much of a substance you have.
Mass is a measure of the amount of matter in an object. The amount of space an object takes up is described by its volume. The density of an object depends on its mass and its volume. Density can be calculated using the following equation:
Density = Mass/Volume
Samples that are the same size but have different densities, will have different masses. Gold has a density of about 19 g/cm3. Pyrite has a density of only about 5 g/cm3. Quartz is even less dense than pyrite and has a density of 2.7 g/cm3. If you picked up a piece of pyrite and a piece of quartz that were the same size, the pyrite would seem almost twice as heavy as the quartz.
Specific gravity is used to identify minerals. Specific gravity measures how heavy the mineral is relative to the same volume of water.
Hardness
Hardness is a mineral’s ability to resist being scratched. Minerals that are not easily scratched are hard. You test the hardness of a mineral by scratching its surface with a mineral of a known hardness. Mineralogists use the Mohs Hardness Scale (Table below) as a reference for mineral hardness. The scale lists common minerals in order of their relative hardness. You can use the minerals in the scale to test the hardness of an unknown mineral.
Mohs Hardness Scale
As you can see, diamond is a 10 on the Mohs Hardness Scale. Diamond is the hardest mineral; no other mineral can scratch a diamond. Quartz is a 7. It can be scratched by topaz, corundum, and diamond. Quartz will scratch minerals that have a lower number on the scale. Fluorite is one. Suppose you had a piece of pure gold. You find that calcite scratches the gold. Gypsum does not. Gypsum has a hardness of 2 and calcite is a 3. That means the hardness of gold is between gypsum and calcite. So the hardness of gold is about 2.5 on the scale. A hardness of 2.5 means that gold is a relatively soft mineral. It is only about as hard as your fingernail.
| Hardness | Mineral |
|---|---|
| 1 | Talc |
| 2 | Gypsum |
| 3 | Calcite |
| 4 | Fluorite |
| 5 | Apatite |
| 6 | Orthoclase feldspar |
| 7 | Quartz |
| 8 | Topaz |
| 9 | Corundum |
| 10 | Diamond |
Cleavage and Fracture
Different types of minerals break apart in their own way. Remember that all minerals are crystals. This means that the atoms in a mineral are arranged in a repeating pattern. This pattern determines how a mineral will break. When you break a mineral, you break chemical bonds. Because of the way the atoms are arranged, some bonds are weaker than other bonds. A mineral is more likely to break where the bonds between the atoms are weaker.
Cleavage
Cleavage is the tendency of a mineral to break along certain planes. When a mineral breaks along a plane, it makes a smooth surface. Minerals with different crystal structures will break or cleave in different ways (Figure below). Halite tends to form cubes with smooth surfaces. Mica tends to form sheets. Fluorite can form octahedrons.
Minerals with different crystal structures have a tendency to break along certain planes. (A) Halite breaks into cubes. (B) Muscovite breaks into sheets. (C) Fluorite breaks into octagons.
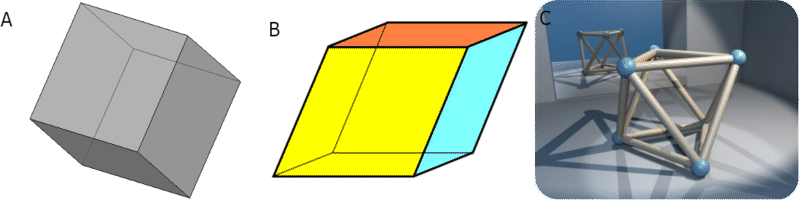
Minerals can form various shapes. Polygons are pictured below (Figure below). The shapes form as the minerals are broken along their cleavage planes. Cleavage planes determine how the crystals can be cut to make smooth surfaces. People who cut gemstones follow cleavage planes. Diamonds and emeralds can be cut to make beautiful gemstones.
(A) Cubes have six sides that are all the same size square. All of the angles in a cube are equal to 90°. (B) Rhombohedra also have six sides, but the sides are diamond-shaped. (C) Octahedra have eight sides that are all shaped like triangles.
Fracture
Fracture describes how a mineral breaks without any pattern. A fracture is uneven. The surface is not smooth and flat. You can learn about a mineral from the way it fractures. If a mineral splinters like wood, it may be fibrous. Some minerals, such as quartz, fracture to form smooth, curved surfaces. Pictured below is a mineral that broke forming a smooth, curved surface (Figure below).
This mineral formed a smooth, curved surface when it fractured.
Other Identifying Characteristics
Minerals have other properties that can be used for identification. For example, a mineral’s shape may indicate its crystal structure. Sometimes crystals are too small to see. Then a mineralogist may use a special instrument to find the crystal structure.
Some minerals have unique properties. These can be used to the minerals. Listed below are some of these properties (Table below); an example of a mineral that has each property is also listed.
| Property | Description | Example of Mineral |
|---|---|---|
| Fluorescence | Mineral glows under ultraviolet light | Fluorite |
| Magnetism | Mineral is attracted to a magnet | Magnetite |
| Radioactivity | Mineral gives off radiation that can be measured with Geiger counter | Uraninite |
| Reactivity | Bubbles form when mineral is exposed to a weak acid | Calcite |
| Smell | Some minerals have a distinctive smell | Sulfur (smells like rotten eggs) |
Summary
- Some minerals have a unique property that makes them fairly easy to identify. But many do not.
- Different minerals can be identified by different properties.
- Cleavage can be a unique and beautiful indicator of mineral type.
Review
- How does streak differ from color?
- How does cleavage differ from fracture?
- List a few minerals and the property that makes each of them unique; e.g. magnetite is magnetic.
Explore More
Use the resource below to answer the questions that follow.
- What color streak would pyrite leave on sandpaper?
- What color streak would real gold leave on sandpaper?
- List some examples of the different types of quartz.
- What characteristic do all types of quartz share?
- What is unique about mica?
- What is mica used for?
- What was azurite used for in ancient times?
- In what colors can calcite be found?
- What color does green calcite streak? Is this true for all colors of calcite?
- What is galena?
- How can you tell that fluorite is not a calcite mineral?
- How many sides does garnet have?